Comments on the Comments on the Greenhouse Gas Effect
In a commentary in late May, Brad Arnold, a reader, provided
commentary on an article that yes, relates to our "favorite" topic: climate change and greenhouse gas emissions.
It wasn't long before Berthold Klein, P.E., took issue with statements in the commentary. Here is the gist of his e-mail:
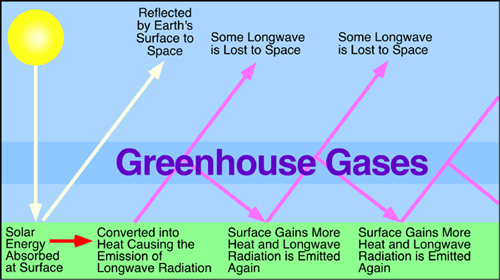
"We all know that the greenhouse effect exists in confined glass spaces, not the atmosphere. The problem is the explanation of why it happens is not understood.
"An experiment proving that the 'greenhouse gas effect exists' must show that there is back radiation caused by properties of CO2 and other gas molecules. It has been shown by Niels Bohr in 1922 that when a gas molecule absorbs electromagnetic energy as photons from infrared radiation, the electrons move to a higher energy shell in the molecule. It does not cause the gas to heat up. The individual molecule can only absorb a specific amount of energy before it is reradiated as 'light energy' with the same wavelength as was absorbed. The light energy will be radiated in any direction. There is no force that causes it to go in the direction that it came from. "Look up the work of R.W. Wood that explains what happens, it is not back radiation of infrared radiation. Another reference that I have given before is 'Greenhouse Gas Hypothesis Violates Fundamentals of Physics' by Dipl-Ing Heinz Thieme. It is a lot easier for the average person to read and understand. It also is consistent to the work of Gerlich etc. Another reference is 'Ponder the Maunder' that is an excellent reading for the beginner. I am not contending that what I have written is absolutely correct, much has been simplified for understanding.
"There is no fixed layer of CO2 in the troposphere. CO2 is distributed throughout the troposphere, thus, if a molecule absorbs specific wavelengths (only 2 or 3 wavelengths in the infrared radiation range), they can be from any direction. During the day, sunlight will be the primary source and after sundown, the CO2 could absorb infrared radiation from Earth in those specific wavelengths, but most of the radiation escapes into space."
Are Klein's comments reasonable to you? Now you can comment directly on our upgraded EP Web site. Please feel free to do so.
Posted by L.K. Williams, EPonline on July 30, 2009