Cleanup Cost-cutter
A guide to the most efficient oxygen-release compound to use in the enhanced bioremediation of contaminated soil and groundwater
- By Bob Kelley
- June 01, 2007
Unfortunately, many of the advances in our industrialized world have come at great expense to our environment. Currently, one of the biggest environmental problems in the United States and other developed nations is the contamination of soil and groundwater caused by accidental releases of hazardous chemicals and petroleum hydrocarbons. Although the U.S. Environmental Protection Agency (EPA) and state environmental agencies have worked to eliminate such releases into the environment, substantial environment contamination has already occurred and needs to be addressed. For example, EPA estimates there are approximately 113,000 underground storage tanks in the United States that have had unintentional releases that need to be cleaned up in order to restore the impacted soils and groundwater.
One of the most successful technologies developed to remediate contaminated sites is bioremediation, which involves using the metabolic activity of microorganisms in order to transform or destroy contaminants. In- situ and ex- situ bioremediation technologies consist of promoting the optimum conditions for naturally occurring indigenous bacteria. With most of these technologies, oxygen delivery is a crucial part of promoting the bacteria’s metabolic activity. Typically, bioremediation is employed to treat soils and groundwater impacted by petroleum hydrocarbons.
The use of injectable, controlled-release, solid-oxygen sources for the enhancement of naturally occurring microorganisms’ metabolic activity has grown exponentially since such products were introduced to the in-situ remediation market in the mid-1990s. But exactly what are these technologies used in bioremediation and how do they work? Why are they so effective, what are the differences between them, and what financial considerations should be made when choosing to use oxygen-release compounds? This article will try to explain how these technologies work and their degree of efficiency in removing contaminants.
Close up on Controlled-Release Compounds
Controlled-release oxygen compounds offer the following benefits when used in the bioremediation of contaminated soils and groundwater: ease of application, no long-term operations and maintenance, environmentally safe, no disposal of hazardous materials, and contaminants are eliminated completely and naturally in the subsurface. In addition, they are cost-effective when compared to other approaches such as pump and treat systems.
One early example of a controlled-release product is the Oxygen Release Compound (ORC®), which was developed in 1994. This fine, white powder, which is 10 percent oxygen by weight, when hydrated produces a controlled release of molecular oxygen into contaminated groundwater and soil to enhance the intrinsically slow rates of natural attenuation. Why the need for oxygen? Without adequate oxygen, petroleum hydrocarbon degradation will either cease or may proceed by much slower anaerobic (oxygen-free) processes. The advantage of this material was that it was easy to apply and could be placed into existing wells in filter socks, or mixed with water and injected as a slurry.
In 2005, a new advanced formulation of this controlled-release product was brought to market as ORC Advanced®. This new controlled-release compound offered the same advantages of the original formulation; however, it was comprised of a form of calcium oxyhydroxide that slowly releases 17 percent oxygen (by weight), thereby producing 70 percent more oxygen per pound than original ORC.
The researchers who developed these two controlled-release compounds noted that in order to stimulate optimally the natural attenuation of aerobically degradable contaminants, biologically usable oxygen is best supplied in low but constant concentrations. The concentration of oxygen in its molecular form (O2) need not exceed 10 milligrams per liter (mg/L) to ensure that the bacteria are satisfied while oxygen losses to atmosphere are minimized. Large bursts of oxygen are wasteful and simply “bubble off,” often generating undesirable foaming and wasting money. If excess oxygen is uncontrollably lost to the atmosphere, inevitably it is not available for biological processes and its source, the oxygen release compound, will have to be replenished. In turn, this needlessly increases both the product and the project costs.
Therefore, manufacturers of these two products engineered them to make use of a patented, controlled-release process. For example, the oxygen release profile of ORC Advanced shows a very controlled, rather constant rate of oxygen release from the time of injection to about 9 months to 12 months depending on site conditions. The release of oxygen has been studied extensively and documented on hundreds of project sites where engineering firms have accumulated data.
Table 1 presents typical field data collected by an engineering firm documenting the longevity of the oxygen release profile for this compound applied on a hydrocarbon-contaminated site. As is clearly shown, the measured dissolved oxygen (DO) slowly increases after a single application of this compound and remains significantly elevated for nine months.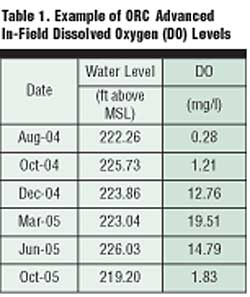
When hydrated, the compound releases oxygen and forms simple calcium hydroxide and water as shown in the following equation:
CaO(OH)2 + H2O ==> ½O2 + Ca(OH)2 + H2O
Each crystal of the compound contains phosphates that retard the hydration of the calcium oxyhydroxide crystal, thereby slowing the formation of oxygen.
Figure 1 shows the comparative oxygen release profile for the advanced controlled-release compound vs. a conventional oxygen release product for the first 75 days of release. Note that after 75 days in the case of the conventional oxygen release product, 80 percent to 90 percent of the oxygen is rapidly and inefficiently released. On the other hand, within the same timeframe the advanced controlled-release compound has released only 21 percent of its oxygen. This comparison clearly demonstrates the advantage between a controlled-release material and a conventional oxygen release material. 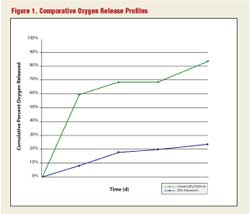
Figure 2 shows the relative efficiencies of treating a plume of contamination in groundwater with the controlled-release compound vs. a conventional oxygen release product. Under typical scenario, the use of commodity product would require three injections. The same project could be accomplished with one injection of the controlled-release compound. This is required because, unlike with the use of the controlled-release compound, the commodity product’s short period of oxygen release will not allow for significant oxygen dispersion from an injection point. Instead, the rapidly released oxygen will simply bubble out of the solution. If one were to use a single initial installation of reinjectable points to avoid the reapplication drilling costs of using the commodity product, then the reinjection points would have to be on much closer spacing than with the controlled-release compound -- on the order of three to four times the number of points in the same area -- to ensure coverage due to low oxygen travel distances associated with the commodity product’s short longevity.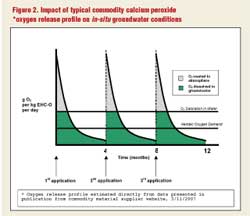
Finding the Right Oxygen Release Compound
One important consideration is the issue of project versus product cost. Often a close look at the cost of a project can reveal areas of inefficiency to target for cost savings. As far as oxygen release compounds go, a clear case can be made for choosing those with controlled-release profiles over short-lived, conventional oxygen release materials.
Comparative costs of using a controlled-release product vs. a noncontrolled release, commodity material are presented in Table 3. This hypothetical situation assumes that the site to be treated requires 1,000 pounds of oxygen to be delivered. Note that while the theoretical material mass required for a controlled-release product and the commodity product are 5,8821bs and 7,143 lbs respectively, this is simply not realistic. Because of the inherent rapid oxygen release characteristics of the conventional product, not much of the oxygen remains in the water, thus requiring quarterly re-injection. In the “realistic scenario” shown in Table 2, the quantity of the commodity material is 14,285 lbs compared to 5,882 lbs for the controlled-release product. This assumes that the second and third injection of the commodity material uses 50 percent of the initial requirement. According to this hypothetical situation, the true cost of using the conventional product is $94,043 vs. $57,840 for the controlled-release product.
Conclusion
By applying injectable, controlled-release solid oxygen source, project managers that use bioremediation to clean up contaminated soil and groundwater can realize good performance and cost savings over traditional, more expensive operations-and-maintenance intensive cleanup approaches, such as pump and treat. The use of per-pound product cost can mask the much lower project costs associated with using controlled-release compounds. Conventional calcium peroxide-based products, although often projecting an image of extended oxygen release, actually cost more due to increased mobilization, product and staffing expenses associated with their required quarterly re-applications.
A Michigan Success Story
Two leaking underground storage tanks (USTs) contaminated soil and groundwater at a service station in Michigan. Project managers at the site used enhanced bioremediation with the advanced controlled-release compound to eliminate an aging and increasingly ineffective pump & treat (P&T) system; this change in treatment technique resulted in the site being closed several years ahead of schedule.
The remediation project began when an environmental site investigation revealed naphthalene, trimethylbenzene (TMB), benzene, toluene, ethylbenzene and xylene (BTEX) contamination in the subsurface prompting the need for remedial cleanup.
The Michigan Department of Environmental Quality (MDEQ) started the cleanup process via UST removal and soil excavation. MDEQ project managers removed a total of 4,000 cubic yards of contaminated soil. They installed a P&T system and operated it through December 2003. The P&T system removed 1,575 pounds of BTEX and significantly lowered contaminant concentrations. However, the system began to lose efficiency as the contaminant mass removed per unit of time decreased drastically.
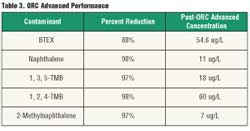
Regulators explored new ways of accelerating the remediation process and reducing the overall cost of cleanup. They decided on enhanced aerobic bioremediation using ORC Advanced to replace the P&T system and facilitate the degradation of the remaining contamination. The remedial plan included two applications of 43 injection points of the controlled-release compound. Three months after the P&T system was shut down, the regulators applied the controlled-release compound on-site. Within 120 days, concentrations were reduced significantly below post-P&T levels (Figure 3) (Reduction continued throughout the monitoring period, and a 99-percent mass reduction was achieved 13 months after the initial injection (Table 3).
|
This article originally appeared in the 06/01/2007 issue of Environmental Protection.
About the Author
Bob Kelley, PhD, is vice president of product development for Regenesis. He has a doctorate in microbiology form Michigan State University. Currently, Dr. Kelley manages all product development activities, which includes improvements and refinements of current products as well as development of new products. His office is in Plainfield, Ill. He can be reached by phone at (815)-531-2705.